Low Pressure Electrolytic Ammonia Production
Creating carbon-free energy from renewable sources.
Current ammonia production technologies are energy intensive and require high levels of heat and pressure to combine nitrogen and hydrogen to create ammonia (NH3), which can then be used as fertilizer; transportation fuel; low carbon, co-combustion additives; and electricity from ammonia fuel cells. The EERC is working to develop low-pressure electrolytic ammonia (LPEA) technology that has the potential to replace the high-pressure Haber Bosch (HB) process, a global standard for NH3 production since 1913. The LPEA production process enables commercially competitive ammonia production in small-scale plants powered by renewable and/or lower-cost off-peak electricity generated at coal/gas-fired utilities.
Ammonia as a Fuel
As fertilizer, NH3 has long been critical to affordable food production. More recently, NH3 is gaining recognition around the world as an economically, environmentally, and strategically valuable fuel because of its 18 weight% hydrogen content, carbon-free composition, storage/transport affordability, and near-zero explosivity risk. Because it contains no carbon, NH3 conversion to energy via fuel cell or combustion generates no CO2 emissions. As a result, if NH3 is produced using renewable energy, water, and air, NH3 becomes a “zero-carbon” fuel, meaning that essentially no CO2 emissions are associated with any aspect of its production and use. Its attributes as a fuel are driving major global investment in technology/infrastructure development.
The LPEA Method
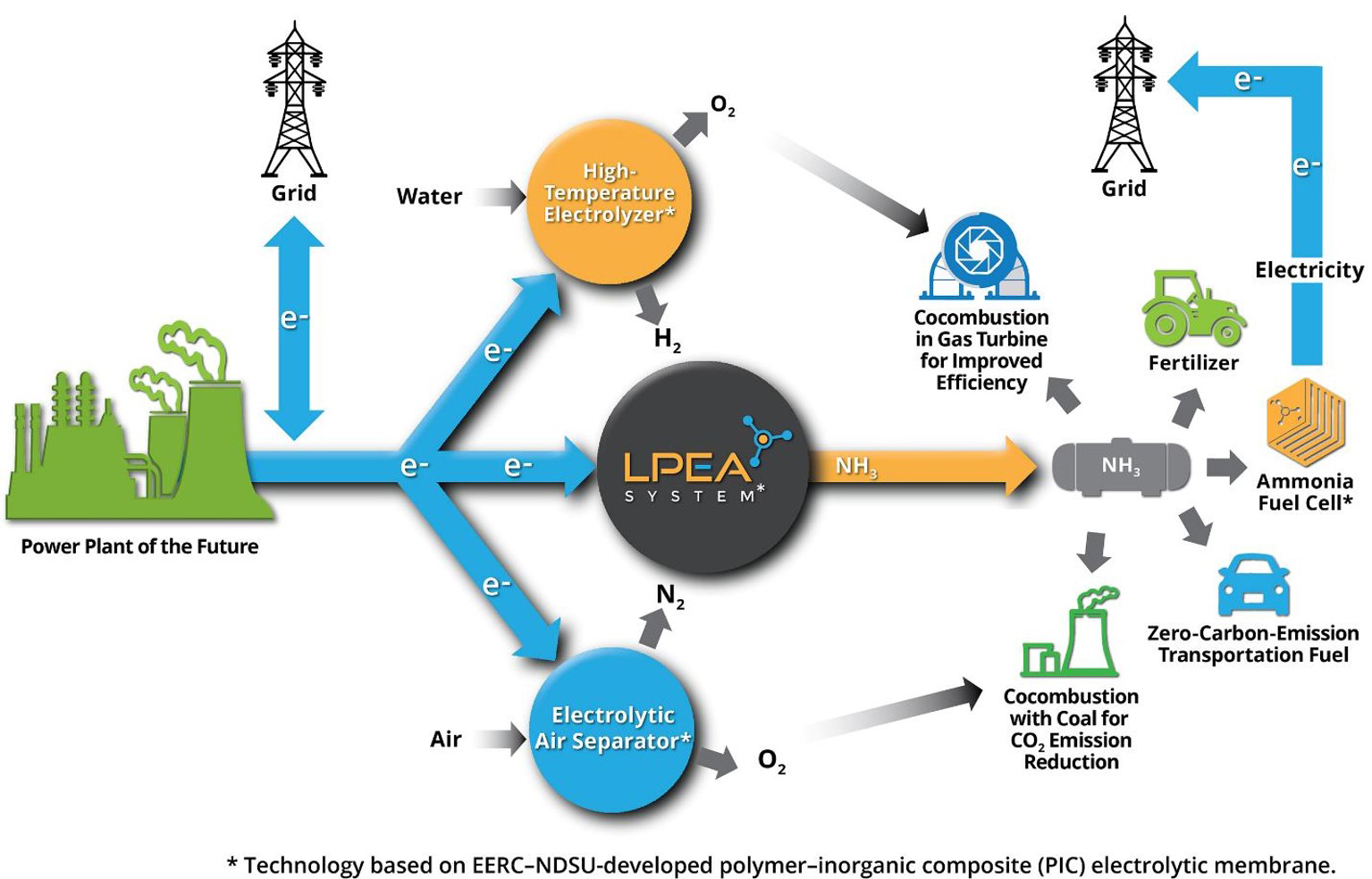
The key enabling technology for LPEA is an innovative EERC-developed proton-exchange electrolyte (PEE) capable of high-rate proton conductance at ≥300°C. The electrolyte comprises small, highly proton-conductive particles distributed in a moldable phosphorous–calcium–sodium (PCN) glass matrix. For NH3 production, an operating temperature of 300–400°C is critical to success, since less than 300°C provides insufficient energy to break the highly stable N2 triple bond, and above 450°C, NH3 decomposes. However, an electrolyte that works at an “intermediate” temperature range of 300–500°C has long been a goal of the international fuel cell/electrolyzer industry.
